Nanotechnology and the COVID-19 Vaccine
Nanotechnology refers to the manipulation of molecules for large-scale manufacturing purposes. It is a broad field of science, which combines chemistry, physics, energy storage, microfabrication, and molecular engineering.
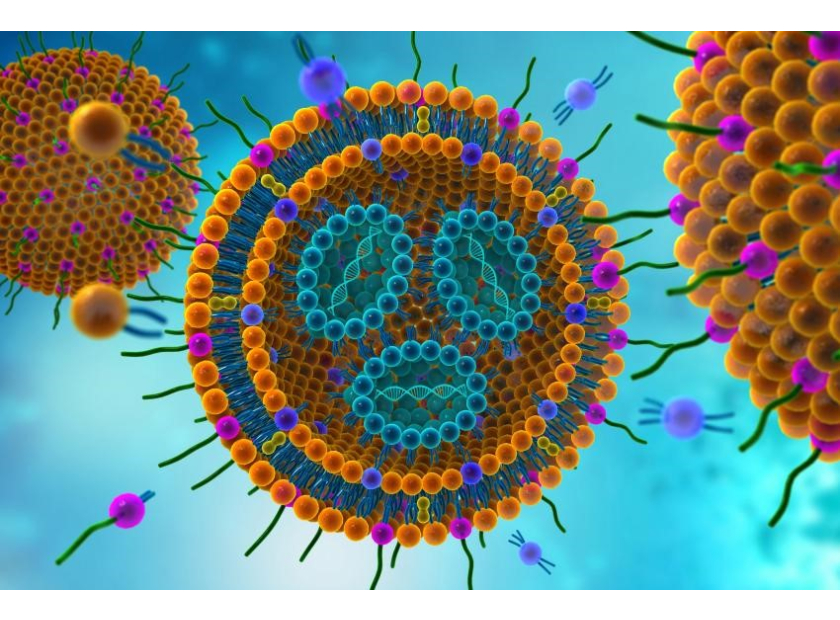
Recently, it has been popularized by its role in nanomedicine for the COVID-19 vaccine. (1) Currently, with the introduction of mRNA vaccines by Pfizer and Moderna, the importance of nanotechnology and the use of liposomes has become even more apparent. Advances in nanotechnology allow for the transport of molecular information, increased protein expression, protection from nucleases, and the improvement of the overall safety of mRNA-based vaccines. (2) Note that the new Johnson & Johnson vaccine uses a modified adenovirus carrier to transport DNA rather than a liposome to transport mRNA.
The primary improvement for mRNA vaccines has to do with the lipid nanoparticle. Lipid nanoparticles (liposomes) are comprised of a phospholipid bilayer, which can be loaded with target drug or mRNA that codes for a target protein, such as those developed for the SARS-CoV-2 vaccine. Although a newer improvement for vaccines, this delivery method has existed for many years and originally gained popularity in 2018 by Onpattro. This was the first drug approved by the FDA to deliver siRNA to treat polyneuropathy. (3) Onpattro offers protection for RNA to avoid host RNAses and, consequently, improves the safe transport of the mRNA to target cells where it is readily absorbed. (2)
The liposomes deliver their mRNA payload into the cell, but the mRNA does not enter the nucleus, nor does it replicate.
Molecular transport is not the only hurdle for mRNA vaccine development. Due to their fragile nature, RNA strands must also go through a strand optimization process. RNA sequences are optimized by adjusting the 5’ and 3’ flanking UTR regions. (2) Alteration of the 5’ cap can be done by several methods and provides more efficient protein synthesis whereas changes to the 3’ poly (A) tail provide greater stability and enhanced translation. (2) In addition, scientists also look at the rarity of codons in the sequence as well and the G:C content for further strand optimization. (2)
With the FDA emergency use authorization (EUA) of the SARS-CoV-2 mRNA vaccines, safety is the main concern for patients. So far, these vaccines have shown to be both safe and effective via large scale clinical trials, after their data was reviewed by the US FDA. (4) The main reason these vaccines are considered safer than other types of vaccines is due to the nature of mRNA’s function within cells. Once mRNA enters the host, it remains in the cytosol until the strand is translated by the ribosomes into a protein product. Soon thereafter, the mRNA transcript is degraded by host enzymes, which reduces the chance of repeated protein production. Furthermore, when the liposome encapsulated mRNA enters the cell in this artificial manner, it lacks the necessary machinery needed to enter the cell nucleus. Thus, new mRNA molecules from the original vaccine source cannot be reproduced. (5)

In conclusion, the use of nanotechnology and liposomal mRNA based vaccines are a promising advancement for future vaccine development. Their current use during the COVID-19 pandemic is showing promising results, and they have the potential for swift modification over other types of vaccines should the need arise with SARS-CoV-2 variants. Their use on a global scale is still new, yet more research and developments in the coming years are being conducted with this technology.
by Miles Amby
R&D Microbiologist
HARDY DIAGNOSTICS