Resistance to β-Lactam Antibiotics: New Solutions to an Old Problem
As we progress through the 21st Century, the looming threat of antimicrobial resistance and the urgent call for action have been expressed by the medical community worldwide. Bacterial resistance mechanisms are becoming increasingly more common, especially to broad spectrum beta (β)-lactam antibiotics which include penicllins, cephalosporins, monobactams, and carbapenems.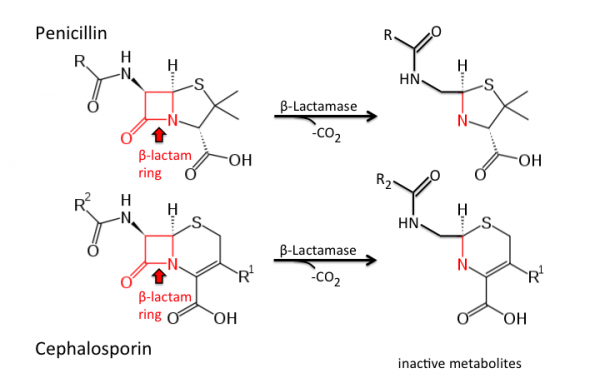
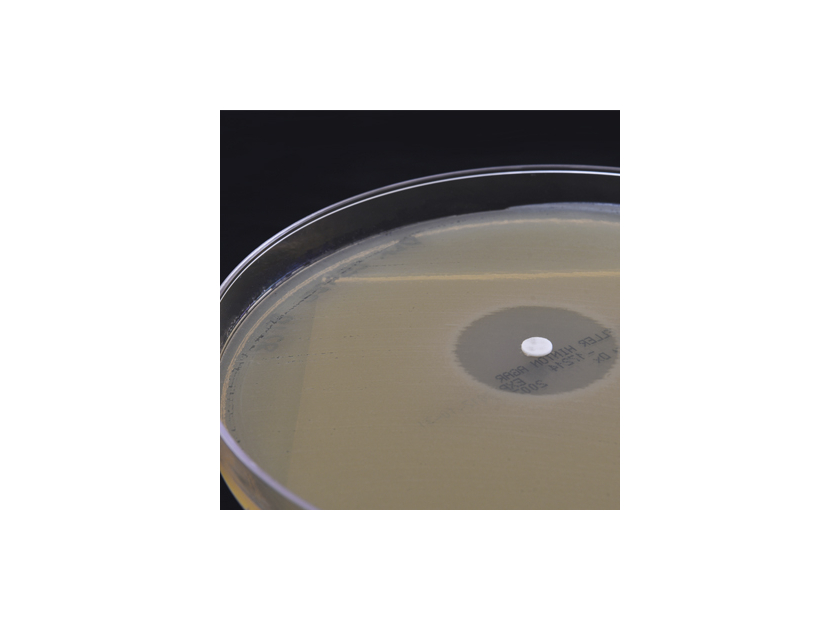
β-lactam resistant bacteria generally evade these antibiotics through two paths: restriction of entry of the antibiotic into the bacterial cell and secretion of β-lactamase enzymes, which neutralize the antibiotic outside of the cell.
FDA cleared pairings of a β-lactam antibiotic and a β-lactamase inhibitor have been widely used in clinical settings for years; however, these combinations are still susceptible to bacterial resistance and may not be viable therapies for future treatment.
Although the recent advancements of new β-lactamase inhibitors is exciting towards fighting antibiotic resistance, older β-lactamase inhibitors are being paired with newer antibiotics and have shown similar effectiveness.
 The β-lactamase inhibitor, tazobactam, discovered in the early 1980s has recently been paired with the 5th generation cephalosporin, ceftolozane, and marketed as Zerbaxa® (Ceftolozane/Tazobactam). This new drug combination is cleared for use to treat complicated urinary tract infections (UTIs) and intro-abdominal infections.
The β-lactamase inhibitor, tazobactam, discovered in the early 1980s has recently been paired with the 5th generation cephalosporin, ceftolozane, and marketed as Zerbaxa® (Ceftolozane/Tazobactam). This new drug combination is cleared for use to treat complicated urinary tract infections (UTIs) and intro-abdominal infections.
Newly discovered β-lactamase inhibitors, such as avibactam and vaborbactam, are showing promising results when paired with conventional β-lactams in treating some of the most antimicrobial resistant strains. For instance, the relatively new FDA cleared β-lactam and β-lactamase inhibitor combinations such as Ceftazidime/Avibactam (AvyCaz®) and Meropenem/Vaborbactam (Vabormere®) have also been effective in treating complicated UTIs and complicated intra-abdominal infections (AvyCaz®). Most importantly, these combination drugs have been successful against many resistant strains harboring extended-spectrum β-lactamases (ESBLs), AmpC β-lactamases, and carbapenemases.
In an article in the Journal of Molecular Microbiology (Calvopiña et al., 2017), researchers have found that although AvyCaz has been shown to be effective clinically, the avibactam does not protect the ceftazidime completely from a particularly resistant β-lactamase producing strain of Stenotrophomonas maltophilia. However, when AvyCaz was paired with aztreonam, a monobactam β-lactam, the inhibition was extremely effective against the S. maltophilia strain. These new β-lactamase inhibitors have not been widely tested and with all β-lactam antibiotics, so their potential effectiveness is still untapped.
Hopefully, this study will be a gateway to further research β -lactamase inhibition and provide new breakthroughs and a potential to reverse antibiotic resistance.
Hardy Diagnostics manufactures many antibiotic disks including the new Ceftazidime/Avibactam (AvyCaz) AST HardyDisks™ for disk diffusion testing. See Cat. nos. Z9351 (pack of 1 cartridge) and Z9355 (pack of 5 cartridges). Meropenem/Vaborbactam (Vabomere) has recently been cleared for use by the FDA and will be available in AST HardyDisks™ in December 2017 (Cat. nos. Z9321 and Z9325).
Another recently launched antibiotic that shows promise is Delafloxacin. While it does not qualify in the β-lactam/β-lactamase category, it is also available in HardyDisk™ (Cat. nos. Z9301 and Z9305).Our Delafloxacin susceptibility disks were also recently cleared for use by the FDA.
References:
- http://www.bristol.ac.uk/news/2017/october/new-hope-in-amr.html
- Karina Calvopiña, Philip Hinchliffe, Jürgen Brem, Kate J. Heesom, Samar Johnson, Ricky Cain, Christopher T. Lohans, Colin W. G. Fishwick, Christopher J. Schofield, James Spencer, Matthew B. Avison. Structural/mechanistic insights into the efficacy of nonclassical β-lactamase inhibitors against extensively drug resistant Stenotrophomonas maltophilia clinical isolates. Molecular Microbiology, 2017; 106 (3):492.
Written by Cameron Whipple (Regulatory Affairs associate) and Andre Hsiung (Director of Technical Services)