USP <71>
Pharmaceutical Sterility Testing
USP <71> is a general testing requirement for accurate, reproducible testing of all pharmaceutical or radiopharmaceuticals that claim to be free from viable organisms. Hardy offers a complete line of all configurations of TSB and Thioglycollate, as well as membrane filtration methods for effective sterility testing.
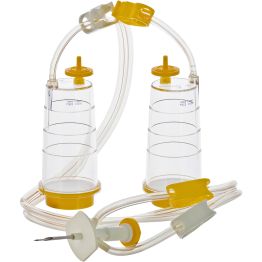
- Sterisart® NF, System with Double Needle, with Septum, 0.45ug, Gamma-sterilized
Cat. no. 16466GSD
PACK SIZE: 10 per package
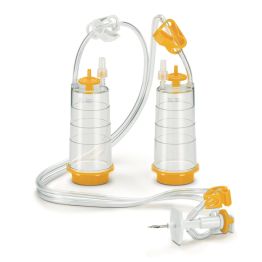
- Sterisart® NF Sterility Test Systems, for Syringes, Sterile, Universal Leur Lock
Cat. no. 16469GSD
PACK SIZE: 10 per package
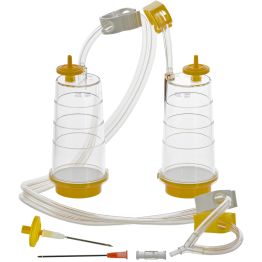
- Sterisart® NF, Short Dual-needle Metal Spike, Sterile, Vented, with Subculturing Septum, 0.45um Membrane
Cat. no. 16476GSD
PACK SIZE: 10 per package
- Sterisart® NF Gamma, System for Difficult-to-dissolve Powders in Unvented Vials, Gamma-sterilized, Sterile, for Use in Isolators, Sterisart® Tubing System
Cat. no. 16470GBD
PACK SIZE: 10 per package