Urogenital Mycoplasmas
Seeing the doctor about concerns of sexually transmitted infections (STIs) or urinary tract infections (UTIs) can be very uncomfortable for most people...
However, it's something that millions of Americans are compelled to do every year. According to the CDC, nearly 2.3 million cases of chlamydia, gonorrhea, and syphilis (the most common STIs) were reported in the US in 2017. This number shows a 31% increase from the 1.8 million reported cases in 2013.
The majority of STI screens
primarily detect Chlamydia trachomatis (chlamydia), Neisseria
gonorrhoeae (gonorrhea), Treponema pallidum subsp. pallidum
(syphilis), Trichomonas vaginalis (trichomoniasis), hepatitis B virus,
and human immunodeficiency virus (HIV). However, another group of bacteria
(Mollicutes) that can cause STIs and UTIs and are often overlooked and not
getting the attention they deserve.
The genital Mollicutes of concern
consist of the bacterial genera Mycoplasma
and Ureaplasma, which are unique
bacteria in that they lack a bacterial cell wall. They are some of the smallest
free-living organisms and can either be pathogenic or commensal in humans. They
have complex nutritional and cultivation requirements that demand special and
laborious techniques for laboratory detection and characterization, which can
discourage clinical microbiology laboratories from performing regular cultures
in-house.
The
reference method for the detection of Mycoplasma
and Ureaplasma is culture-based,
typically on A8 agar. Nevertheless, due to the insensitivity of culture,
requirement of specific media and the length of the culture (typically a few
days to a couple of weeks for colony maturation), the optimal detection method
is PCR-based. However, in the clinical setting, detecting these urogenital
mycoplasmas is rarely urgent, so labs may elect to not use PCR because of this
and the high costs associated.
Historically, these organisms have
not been studied closely due to their fastidious nature and small size.
Traditionally, they are not considered nor screened for in the initial STI or
UTI battery of tests. However, Mycoplasma
hominis and Ureaplasma urealyticum can
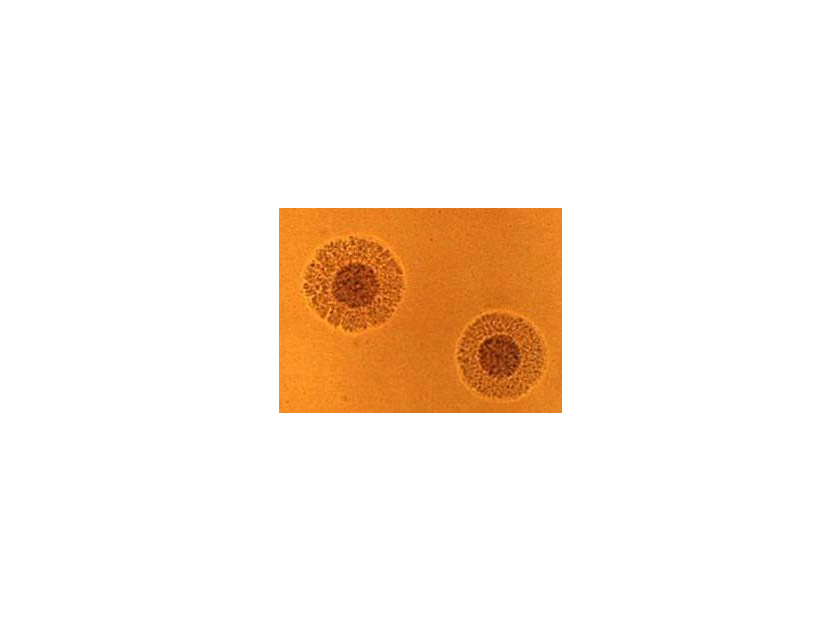
cause some serious infections and conditions if misidentified. Microscopically,
on A8 agar, Mycoplasma hominis mimic
a “fried-egg” morphology (see below).

M. hominis has been isolated
from the upper urinary tract in patients with acute pyelonephritis, a kidney
infection that causes pain and inflammation. M. hominis infection during
pregnancy can cause ectopic pregnancy, early delivery, or miscarriage, and can
induce fever among newborns. U. urealyticum causes non-chlamydial,
non-gonococcal urethritis (NGU). In newborns, an U. urealyticum
infection during pregnancy can cause low birth weight, pneumonia, and
septicemia.
These microorganisms are typically
transmitted via sexual contact or vertical transmission (mother to child) and
may go seemingly unnoticed depending on the patient's immune system
health. Although M. hominis and U. urealyticum can be
present in the commensal flora of many humans, when they exceed concentrations
of 104 and 103 CCU/mL, respectively, these bacteria can
often lead to urogenital tract infections.
Although these bacteria can exist
in the commensal flora of many humans, their unmonitored proliferation can
prove pathogenic. It is quite common that when one is suffering from STI or
UTI-related symptoms, a Mycoplasma
test is often the last test ordered by the clinician, only after all other
pathogen possibilities haven been exhausted and all laboratory results return
negative. Beta-lactams (including penicillins and cephalosporins) are
ineffective against these organisms because they lack a cell wall. Therefore,
doxycycline, macrolides (eg. azithromycin), and fluoroquinolones are often used
to treat these infections.
Luckily, there is a device that can
detect, identify, and enumerate Mycoplasma
hominis and Ureaplasma urealyticum
from endocervical, urethral, urinary, gastric and sperm specimens in as little
as 24 hours. This device is called Mycofast US by ELITech and is now for sale
in the US by Hardy Diagnostics. This “all liquid”, rapid colorimetric test
relies on innate properties of M. hominis
and U. urealyticum for detection and
identification, and patented techniques for enumeration. The color pattern of
wells in the device is used to interpret the results of the test. A positive
result indicates that Mycoplasma hominis and/or
Ureaplasma urealyticum are present in
the specimen tested, either colonization or infection. However, the positive
test alone should not be used to make a clinical diagnosis. The diagnosis
should be determined by the physician based on the correlation between the
culture results and the clinical symptoms present.
This test is a great compromise between the traditional Mycoplasma culture and PCR methods because of two main characteristics: 1) Time – results can be achieved in as little as 24 hours, 48 hours for strains with weaker enzymatic activity, which is considerably quicker than the weeks required for typical cultivation and 2) Money – this test is less expensive than PCR-based detection methods. Order your very own box of 30 MYCOFAST US tests (cat. no. 00050) at hardydiagnostics.com today.

by Dylan Campbell and Andre Hsiung