Featured Posts
-
Posted: December 26, 2024Read More
Introduction to Antimicrobial Resistance (AMR)
Antimicrobial resistance (AMR) is one of the top ten global public health threats facing humanity.¹ Ironically, humans are to blame; yet, in recent years, the increase of AMR strains in animals, specifically companion animals, has also...
-
Posted: December 20, 2024Read More
Two hundred years ago, give or take the odd decade, Pseudomonas aeruginosa was an environmental bacterium,¹ apparently not one, as far as medical records in the pre-microbiology days can be discerned, associated as a human pathogen.²
Today, P. aeruginosa is associated with a high number of multidrug-resistant infections,³...
-
Posted: December 09, 2024Read More
Cleanrooms and the protection of product carry two risks from contamination: particle deposition and particle dispersion. Airborne particle contamination in the healthcare and pharmaceutical sectors is a major topic of concern due to the risk of introducing particles, as impurities into products, and due to the potential to transfer microorganisms. Air quality assessments are partly based on the amount...
-
Posted: November 26, 2024Read More
The cold and flu season is upon us.
Flu season tends to peak in the winter months, between December and February, according to the Centers for Disease Control and Prevention. If you have not stocked up yet on necessary testing supplies for your laboratory, we are listing a few key...
-
Posted: November 20, 2024Read More
It's been one year since the revised United States Pharmacopeia Chapter <797> was implemented. Over the course of the year, we have received feedback that many teams have faced challenges with the increase in environmental monitoring requirements and frustration over how this has affected their facility. Many pharmacies contract with third-party vendors to conduct their air sampling and other...
-
Posted: October 14, 2024Read More
Recent studies have shown that many tattoo inks contain dangerous chemicals and potentially infectious bacteria. Despite their cultural popularity and potential hazards, the U. S. Food and Drug Administration (FDA) does not regulate the tattoo industry. The FDA considers tattoos and permanent makeup as cosmetics, so they do not regulate inks used for the purpose of injection into skin; they do not...
-
Posted: October 11, 2024Read More
In December of 2022, President Biden signed the Modernization of Cosmetics Regulation Act (MoCRA) into law, ushering in new regulations that significantly expanded the authority of the FDA over personal care companies. There have been years-long efforts to increase the FDA's oversight of the personal care and cosmetic industry, stemming from growing concerns regarding product safety. In 2018, there...
-
Posted: October 08, 2024Read More
Carbapenem-resistant Enterobacterales (CRE) refers to a group of bacteria within the Enterobacterales order that have developed resistance to carbapenem antibiotics. Carbapenems are a class of broad-spectrum antibiotics used as a last-resort treatment for serious infections caused by multi-drug-resistant bacteria.
... -
Posted: September 16, 2024Read More
When performing Growth Promotion on new batches of media, one of three plating techniques can be used: Spread Plate, Pour Plate, or Membrane Filtration. Each method aids in enumeration of the cells in the inoculum, but each is different in technique. Below we offer our Top 5 Best Practices for each plating method to help you ensure proper growth promotion of your agar media...
-
Posted: September 12, 2024Comments: 1Read More
Globally, millions of individuals are affected by UTIs each year, of which, a portion of these cases may be caused by consuming ExPEC from food reservoirs. Additional studies are necessary to clarify the relationship between UTI risk and the presence of ExPECs in food reservoirs. It is important for researchers to identify the level of risk ExPECs pose to human health...
-
Posted: September 09, 2024Read More
The birthrate in the United States has hit a new record low, according to an analysis of 2023 birth certificate data published this past spring by the Centers for Disease Control and Prevention (CDC). Last year’s slowdown marks an end to the uptick in new babies that began during...
-
Posted: August 06, 2024Read More
Explore the remarkable life of Sir Alexander Fleming, the man behind penicillin, in our latest blog post. Born in 1881 in Scotland, Fleming's journey from a marksman in the Territorial Army to a Nobel laureate revolutionized modern medicine. Discover his accidental discovery of penicillin and its profound impact on global health. Dive into the history and personal stories that shaped one of the greatest...
-
Posted: July 08, 2024Read More
The award from Communify of Santa Barbara County is a significant milestone for Hardy Diagnostics and a testament to the incredible work of our CAC. We are grateful for the recognition and inspired to continue our mission of improving lives both inside and outside the laboratory. Congratulations to Lauren Hamilton and the entire committee for their outstanding contributions...
-
Posted: June 03, 2024Read More
Wright-Giemsa Stains, derived from the once groundbreaking Romanowsky stains, are stains used in hematology and cytology studies to differentiate cells in microscopic examinations of peripheral blood and bone marrow specimens. Review our latest tutorial to learn more...
-
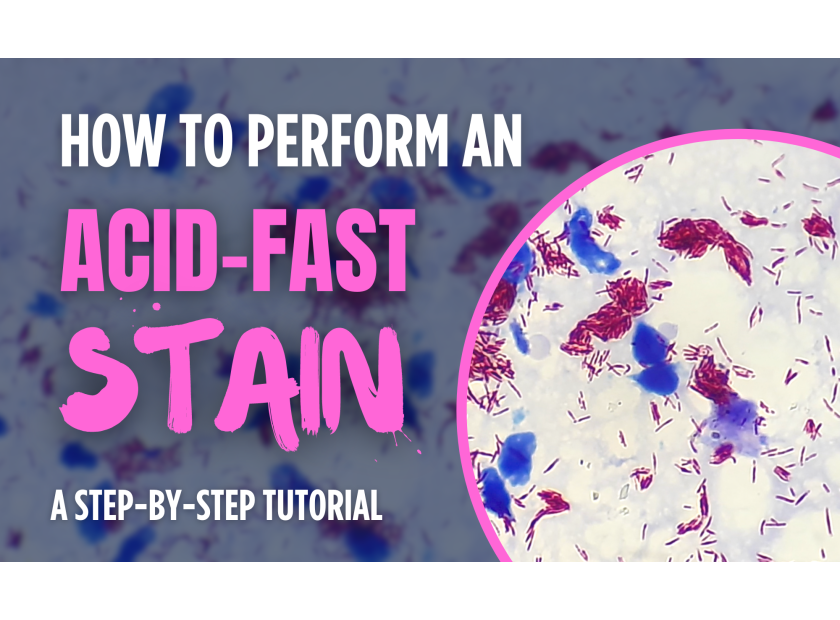
Posted: May 28, 2024Read More
Performing an acid-fast stain is a crucial technique in the field of microbiology, especially when it comes to identifying mycobacteria, such as Mycobacterium tuberculosis, the causative agent of tuberculosis. This staining method distinguishes acid-fast bacteria from non-acid-fast bacteria based on the unique properties of their cell walls. In this tutorial, we will guide you through the step-by-step...