Antibody-Dependent Enhancement: A Critical Challenge in Vaccine Safety
Our immune system plays a significant role in keeping us alive. As a first line of defense, a non-specific innate immune response is triggered directly by pathogens. Subsequently, more sophisticated and specific responses, known as adaptive immune responses, are called into action. Both work together to detect and eradicate the invading pathogens.1,2 Every multicellular organism must safeguard itself against potential threats like pathogens; failure to do so could result in its swift demise.1
A primary objective in the development of vaccines and therapeutics is to stimulate the body to produce antibodies that block pathogen entry into cells and tissues. Vaccines emulate infections by presenting an antigen to our immune system. This antigen may be a weakened or inactivated virus or bacteria, components of their outer surface, or genetic material. As part of the adaptive immune response, our bodies generate neutralizing antibodies that attach to pathogens, hindering their ability to infiltrate cells. However, on rare occasions, antibodies can act as a Trojan horse that facilitates the pathogen’s access into cells and potentially worsen the disease.2,3,4
Some antibody inducing vaccines and their adjuvants carry the risk of aggravating disease through the mechanism of antibody-dependent enhancement (ADE).3 When ADE is triggered, these antibodies can enhance virus entry and replication in the cells. Historically, several viruses including the dengue virus, respiratory syncytial virus (RSV), human immunodeficiency virus, coronaviruses, and measles have been found to demonstrate ADE effects.5 Some researchers have proposed that this may be the case with SARS-CoV-2 as well.3,5,6
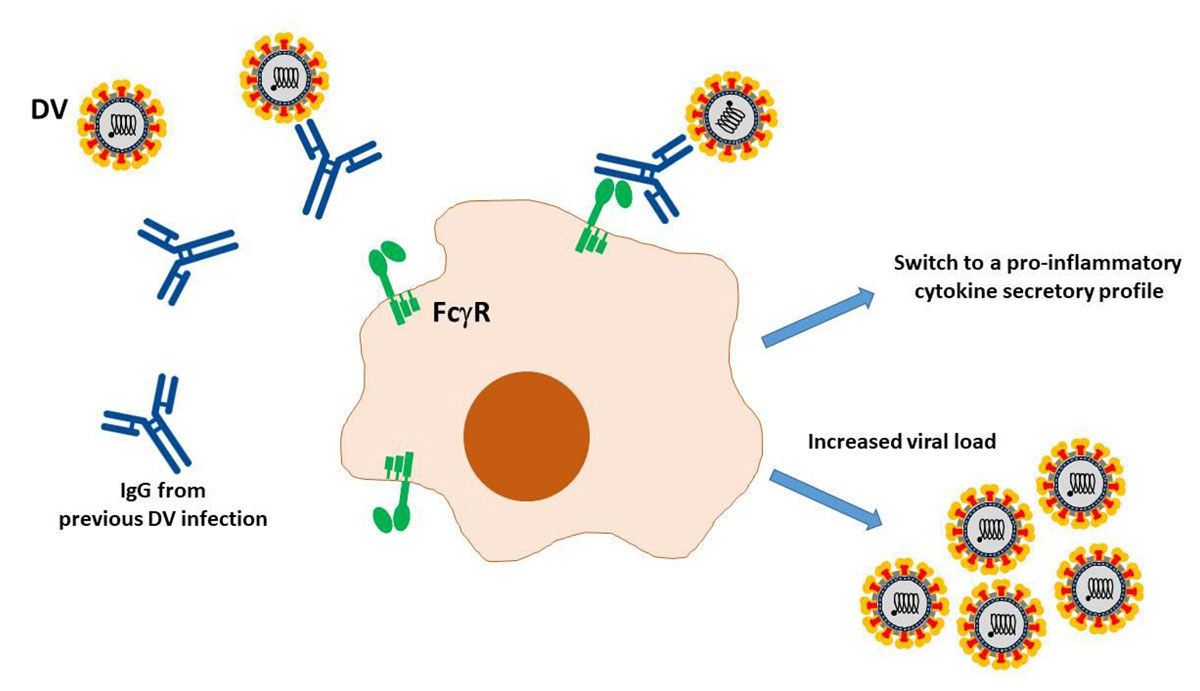
RSV, recognized for its propensity to cause pneumonia in children, prompted the development of an antibody-based vaccine utilizing inactivated virus as an antigen. However, during clinical trials, children who received the vaccine exhibited higher rates of pneumonia and even death after RSV infection. Due to these alarming findings, the vaccine trials were stopped, and the vaccine was never approved or released to the public.2
The early iteration of the measles vaccine, produced by formaldehyde inactivation of the measles virus, yielded similarly concerning outcomes. Vaccinated children who subsequently contracted measles exhibited severe symptoms, including high fevers, distinct rashes, and atypical pneumonia. Prompted by these adverse effects, the vaccine was discontinued. Recipients of this vaccine were advised to undergo re-vaccination with the live, attenuated measles vaccine, which does not induce ADE, and remains the standard vaccine in use today.2
While acknowledging the potential risk of vaccine-induced ADE, it is imperative to emphasize that modern vaccine formulations undergo rigorous scrutiny through comprehensive clinical trials as a prerequisite for regulatory approval. These rigorous evaluations serve to mitigate the likelihood of ADE occurrence following vaccination.2
Meet the author

R&D MICROBIOLOGIST II at HARDY DIAGNOSTICS
Geovanni Ruiz, B.S. Biological Science
Geovanni graduated from San Jose State University in 2020 with a degree in Biological Science and minor in Chemistry. His professional journey began as an Analytical Chemist at an esteemed environmental and oilfield testing laboratory, where he meticulously operated a variety of instruments to detect metals in solid and water samples. Seeking to broaden his expertise, Geovanni transitioned to a Microbiologist position at a prominent food testing laboratory. Here, he applied his knowledge to analyze food and beverage samples for the presence of microorganisms. In November 2023, Geovanni joined Hardy Diagnostics as an R&D Microbiologist II, marking a significant step forward in his career. In this role, he spearheads new product development initiatives, troubleshoots existing product issues, evaluates raw materials, and oversees small-scale pilot batches. Outside of his professional endeavors, Geovanni is passionate about music, playing guitar in his leisure time. He finds solace and inspiration in gardening, enjoys dancing as a form of creative expression, and finds adventure in mountain biking. Geovanni's dedication to scientific excellence, combined with his multidisciplinary background and creative pursuits, positions him as a valuable asset in the field of microbiology and beyond.